 
Use whichever seems more obvious to you while you are trying to understand this, but for exam purposes learn whichever is in your syllabus. 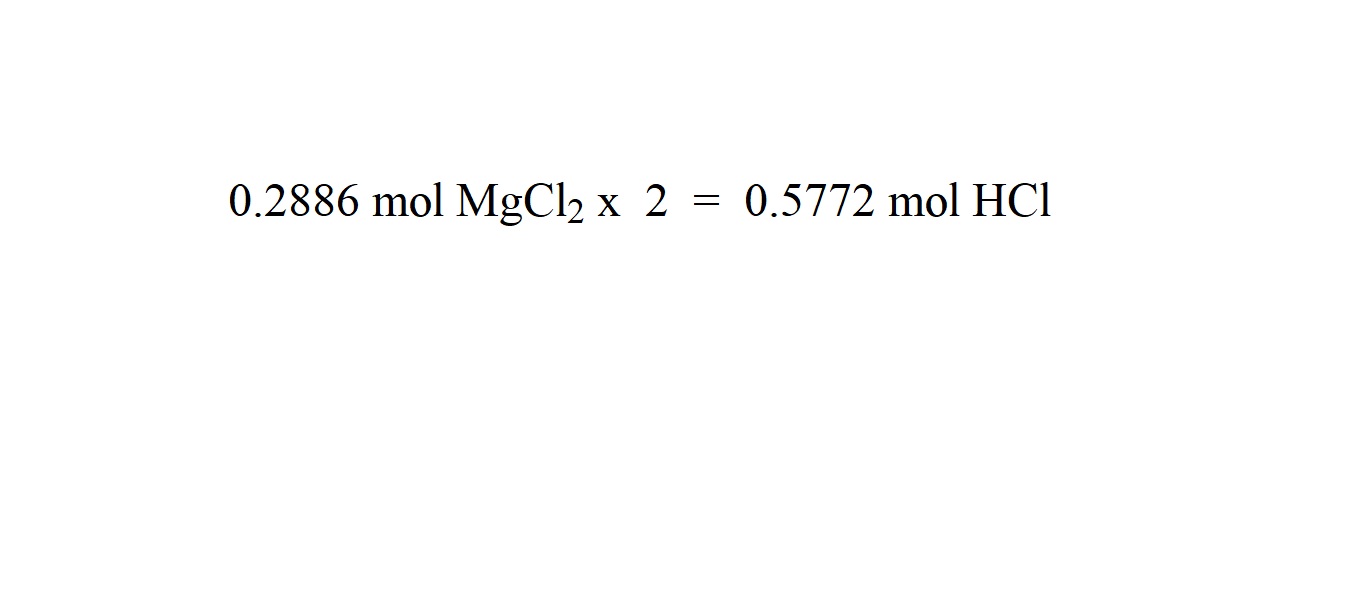
The relative isotopic mass is the mass of the isotope on a scale on which the mass of a carbon-12 atom is exactly 12 units.įor example, an atom of Mg-24 is twice as heavy as an atom of C-12, and so is given a relative isotopic mass of 24. There is an alternative phrasing for this which some people find easier to understand. The relative isotopic mass is the mass of the isotope relative to 1/12 of the mass of a carbon-12 atom. Most people start with relative atomic mass but it makes sense to talk about relative isotopic mass first. Weighing atoms in a standard mass unit like grams would be daft! For example, it would take about 6 x 10 23 hydrogen atoms to weigh 1 g. You can't use a familiar mass unit like a gram because atoms are so small. The masses of atoms are measured relative to the mass of a C-12 atom. They have the same number of protons but different numbers of neutrons. Isotopes are atoms which have the same atomic number but different mass numbers. The fact that they have varying numbers of neutrons makes no difference whatsoever to the chemical reactions of the carbon. These different atoms of carbon are called isotopes. They all have the same number of protons, but the number of neutrons varies. For example, there are three kinds of carbon atom 12C, 13C and 14C. The number of neutrons in an atom can vary within small limits. The importance of the C-12 isotope in chemistry calculations This page explains the atomic mass scale based on the mass of an atom of the C-12 isotope. Relative atomic mass, relative molecular mass and relative formula mass Relative atomic mass and relative formula mass 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
